
The Bioanalytical Services in Ensuring Drug Safety and Efficacy
In the process of bringing a drug from the laboratory to the patient, every stage undergoes thorough scrutiny, highlighting the
Follow Us On:

In the process of bringing a drug from the laboratory to the patient, every stage undergoes thorough scrutiny, highlighting the

Biostatistics plays a crucial role in the healthcare and life sciences sectors, and biostatistics services apply statistical principles to address

Data Management Systems play a pivotal role in the high-stakes realm of pharmaceutical development, where the urgency to bring a

In the fast-evolving world of healthcare and pharmaceuticals, clear and compliant communication is not just a necessity—it’s a strategic advantage.
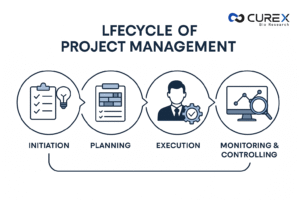
Clinical research relies heavily on effective clinical project management for the successful execution of trials. The lifecycle, outlined in the

In the ever-evolving landscape of clinical trials, site monitoring services play a pivotal role in safeguarding data quality, patient safety,
Pharmacovigilance (PV) is crucial in the fast-paced pharmaceutical development world, as it ensures drug safety and human lives. CUREXBIO is

A well-designed clinical development plan (CDP) is crucial in drug development, guiding pharmaceutical companies from preclinical testing to regulatory approval.

In today’s fast-evolving landscape of clinical research, where digital platforms and remote operations are becoming the norm, prioritizing compliance and

1113, Silver Radiance 4 Sarkhej – Gandhinagar Hwy, Chanakyapuri, Ahmedabad, Gujarat 382481

7851 SW Ellipse Way Suite 16 Stuart, Florida 34997

203 Ziadodda Cresent, Brampton,Ontario, L6P1T4
© Copyright 2025 Curexbio. All Rights Reserved