
The Critical Role of Central Laboratory Support in Biomarker-Driven Clinical Trials
The era of one-size-fits-all medicine has ended, giving way to biomarker-driven clinical trials that represent the forefront of precision medicine.
Follow Us On:

The era of one-size-fits-all medicine has ended, giving way to biomarker-driven clinical trials that represent the forefront of precision medicine.

Clinical data management is vital in clinical trials, guaranteeing that data is accurate, reliable, and prepared for analysis and regulatory

In the process of introducing a new therapy, the Clinical Study Reports (CSR) serves as the key scientific, clinical, and

The landscape of medical writing is rapidly transforming due to artificial intelligence (AI), financial pressures, and changing global regulations. In

Selecting an appropriate Clinical Research Organization (CRO) is crucial in drug development, particularly within India’s expanding clinical research market. This

In the dynamic realm of pharmaceutical and biotechnology innovation, the journey from drug or therapy concept to market involves intricate

Clinical trial data management (CDM) is essential in pharmaceutical and biotechnology research, enabling the effective handling of extensive data from
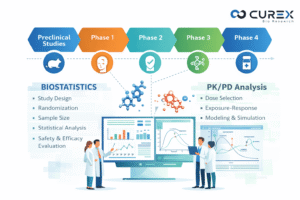
Biostatistics services play a pivotal role in drug development, where the transition from a promising molecule to an approved therapy

In the rapidly evolving pharmaceutical sector, the safety profile of a drug continually develops post-regulatory approval, necessitating ongoing signal detection

1113, Silver Radiance 4 Sarkhej – Gandhinagar Hwy, Chanakyapuri, Ahmedabad, Gujarat 382481

7851 SW Ellipse Way Suite 16 Stuart, Florida 34997

203 Ziadodda Cresent, Brampton,Ontario, L6P1T4
© Copyright 2025 Curexbio. All Rights Reserved