
Regulatory Affairs Pre-Submission Checklist: Building Bulletproof Protocols for Regulatory Success
If you work in the biotech or pharmaceutical sectors, you understand that an inadequately structured protocol can significantly hinder a
Follow Us On:

If you work in the biotech or pharmaceutical sectors, you understand that an inadequately structured protocol can significantly hinder a
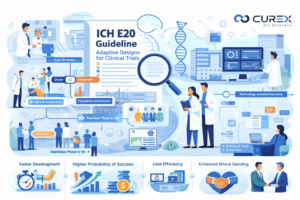
The clinical development landscape is experiencing significant changes, moving away from traditional, linear, and fixed Clinical Trials designs. These conventional

In the competitive arena of clinical development, the urgency to expedite the introduction of new therapies is paramount. Sponsors face

In clinical research, data integrity serves as the cornerstone for conclusions, regulatory submissions, and patient safety. While routine checks and

In clinical trials, data integrity is crucial, yet even meticulously designed studies can encounter issues due to inconsistencies or gaps

In the high-stakes arena of drug development, data has emerged as a vital asset. The extensive process of moving from

1113, Silver Radiance 4 Sarkhej – Gandhinagar Hwy, Chanakyapuri, Ahmedabad, Gujarat 382481

7851 SW Ellipse Way Suite 16 Stuart, Florida 34997

203 Ziadodda Cresent, Brampton,Ontario, L6P1T4
© Copyright 2025 Curexbio. All Rights Reserved