Pharmaceutical products approved for human use promise to heal, alleviate, or prevent disease, but they also carry risks. The balance between benefits and harms requires constant vigilance, which is provided by pharmacovigilance—the science dedicated to detecting, assessing, understanding, and preventing adverse drug effects. This discipline acts as an early warning network in the healthcare system, ensuring that patient medication use is monitored and analyzed for safety. Modern pharmacovigilance is founded on two key components: signal detection and risk management. Signal detection uncovers potential safety issues within extensive data, while risk management addresses and communicates these concerns systematically to minimize risks.
CurexBio has developed its pharmacovigilance practice based on years of regulatory expertise. The company offers comprehensive pharmacovigilance services throughout the drug safety lifecycle, including case safety report processing, signal detection, and risk management plan development. This blog highlights the importance of signal detection and risk management in drug safety, emphasizing how CurexBio’s expertise assists sponsors in navigating this complex arena.
Table of Contents
ToggleUnderstanding Pharmacovigilance for Drug Safety
The term pharmacovigilance originates from the Greek pharmakon (drug) and Latin vigilare (to keep awake or alert), denoting the discipline focused on remaining vigilant about drug safety.
Every pharmaceutical product approved for market use has benefits but also adverse effects. Timely detection of unknown risks is crucial for patient safety, applying throughout the entire life cycle of medicinal products from clinical development to post-market use.
The function’s importance is highlighted by history, which has shown the severe consequences of insufficient pharmacovigilance.
The Thalidomide Tragedy (1961): Thalidomide use during pregnancy resulted in thousands of babies being born with severe congenital abnormalities, leading to a fundamental shift in pharmaceutical safety regulation globally.
The Sulphanilamide Elixir Tragedy (1937): A toxic solvent, diethylene glycol, in an elixir resulted in over 100 deaths in the United States, prompting modern drug safety legislation.
This incident highlighted the importance of reporting, leading to today’s pharmacovigilance systems intended to prevent future tragedies.
What Is Signal Detection?
- Signal detection is the vital first step in pharmacovigilance, focused on identifying potential safety signals from diverse data sources for further investigation.
- A safety signal indicates information from one or more sources that implies a new or revised causal link between an intervention and an event, whether adverse or beneficial, warranting further investigation.
- Signal detection in drug safety acts like a radar system, continuously monitoring for potential issues by identifying patterns.
The Signal Management Process
Signal management consists of a structured five-phase process aimed at systematically identifying, evaluating, and addressing potential safety concerns.
| Phases | Role | Objective |
| Signal Detection | Regular screening of adverse event databases, such as VigiBase, FAERS, and EudraVigilance, involves employing data mining techniques and clinical assessments to identify potential safety concerns associated with medical products. | Identify potential medicine–event combinations requiring investigation |
| Signal Validation | Checking whether the adverse effect is already covered in product information; excluding other causes | Confirm the combination warrants further assessment |
| Signal Prioritisation | Ranking validated signals based on seriousness and public health impact | Allocate resources to most critical signals first |
| Signal Assessment | Evaluating causal relationship using tools like Bradford Hill criteria; reviewing scientific literature | Determine whether evidence supports a causal association |
| Signal Communication | Disseminating findings to regulators, marketing authorization holders, and public | | Enable appropriate regulatory action |
Signal Detection Methods
Modern signal detection employs both qualitative and quantitative approaches, often in combination .
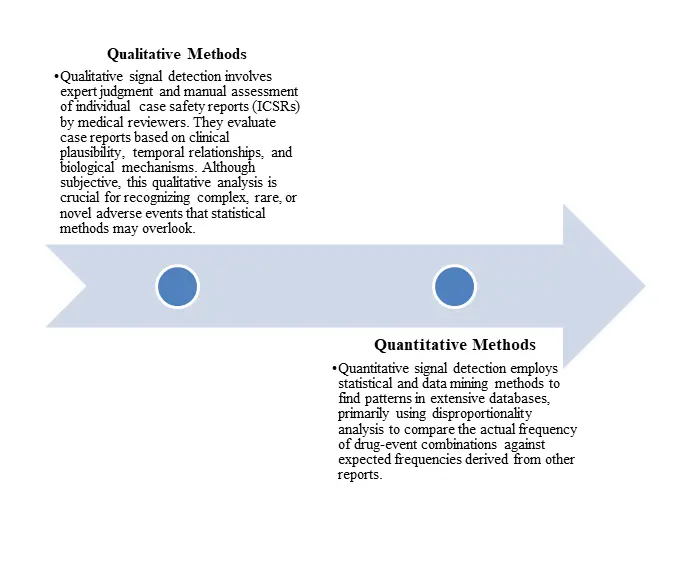
Key quantitative measures include :

These statistical methods efficiently screen large datasets to identify potential signals for expert clinical review.
Types of Signals
| Signal Type | Description |
| Spontaneous Reporting Signals | | Derived from unsolicited reports from healthcare professionals and patients |
| Data Mining Signals | Identified through statistical analysis of large databases |
| Literature-Based Signals | Emerging from systematic reviews, meta-analyses, and case reports |
| Prescription Event Monitoring (PEM) Signals | Tracking specific drugs in patient cohorts |
| Periodic Safety Update Report (PSUR) Signals | Identified during routine safety reporting |
| Registry Signals | Long-term safety data from patient registries |
Data Sources for Signal Detection
The FDA’s Sentinel System showcases a sophisticated signal detection infrastructure that leverages real-world data from over 300 million individuals in the U.S. to provide evidence regarding adverse drug reactions, thus enhancing traditional spontaneous reporting with its “active surveillance” capability.

Risk Management: From Signal to Action
Risk management is crucial after a signal has been detected, validated, and assessed, as it determines the actions to be taken in response.
What Is a Risk Management Plan (RMP)?
A Risk Management Plan (RMP) is a strategic document required by regulatory authorities that details how a pharmaceutical company will monitor, assess, and mitigate risks linked to a medicinal product, ensuring a positive benefit-risk profile throughout its lifecycle.
RMPs encompass identified and potential risks, as well as any incomplete safety information that necessitates further investigation post-approval. Their fundamental objective is to deliver a thorough strategy aimed at enhancing patient safety by reducing adverse effects and optimizing therapeutic efficacy.
Key Components of a Risk Management Plan
| Component | Purpose | What It Includes |
| Safety Specification | Characterizes the safety profile, concentrating on important identified and potential risks and missing information | Clinical trial findings, preclinical data, spontaneous reports, literature; categorization into identified risks, potential risks, missing information |
| Pharmacovigilance Plan | Outlines how safety will be monitored post-approval | Routine activities (spontaneous reporting, PSURs, data mining) and additional activities (PASS studies, active surveillance, registries) |
| Evaluation of Risk Interventions | Assesses effectiveness of implemented safety actions | Routine measures (labeling, SmPC updates) and additional measures (physician training, controlled distribution, educational materials) |
| Risk Minimisation Plan | Describes actions to prevent or reduce the probability and/or severity of adverse reactions | Routine measures (labeling, SmPC updates) and additional measures (physician training, controlled distribution, educational materials) |
When Is an RMP Required?
An RMP is required for all new marketing authorization applications involving innovative drugs, biologics, biosimilars, and advanced therapy medicinal products, irrespective of the therapeutic area.
Additional situations requiring RMP submission or updates include :
- Significant changes to existing marketing authorizations (MA) include the introduction of new dosage forms, new routes of administration, and new manufacturing processes for biotechnological products, as well as substantial indication changes such as new disease areas and age groups, including pediatrics.
- Regulatory requests arise when the EMA or national authorities detect issues impacting the benefit-risk balance of a product.
- Renewal of Marketing Authorizations (MA) is required for products that already have Risk Management Plans (RMPs). This process ensures that the RMPs are updated and compliant with current regulations.
- Significant changes to the benefit-risk balance occur when there are new safety concerns or major formulation changes, however, generic products and well-established medicines are usually exempt from this unless such concerns arise.
Routine vs. Additional Pharmacovigilance Activities
The pharmacovigilance plan within a Risk Management Plan (RMP) distinguishes between various aspects relevant to drug safety monitoring and reporting.
- Routine Activities (Passive Surveillance):**
- Spontaneous adverse event reporting and analysis
- Periodic Safety Update Reports (PSURs) compiling event data over specific periods
- Routine data mining of safety databases
- Signal detection activities
Additional Activities (Product-Specific):
- Non-clinical studies to understand toxicity mechanisms
- Pharmacogenetic studies investigating genetic influences on response
- Stimulated reporting through patient organizations
- Active surveillance (pregnancy follow-up, registries)
- Non-interventional/observational studies (prospective cohort studies)
- Clinical trials for safety evaluation
Real-World Examples of Risk Management
- Quinolone antibiotics have been linked to tendon rupture primarily through post-marketing reports rather than clinical trials. Initially considered biologically implausible by many clinicians, the growing number of case reports resulted in boxed warnings for all quinolone labels. This situation underscores the importance of ongoing surveillance, as serious but rare risks may only surface after extensive use.
- Troglitazone, a diabetes medication, was withdrawn due to hepatic toxicity cases detected in post-marketing surveillance. Identifying such rare hepatic reactions requires a trial size of about 30,000 patients, which exceeds typical pre-approval databases, making post-marketing surveillance essential for risk identification.
The Future of Pharmacovigilance: AI and Innovation
- AI-Driven Signal Detection
Artificial intelligence is transforming pharmacovigilance by improving signal detection through sophisticated algorithms and data analysis. AI’s capability to rapidly process large datasets and recognize intricate patterns enhances the prediction of safety issues more effectively than traditional approaches. The FDA’s Sentinel Innovation Center is exploring machine learning and natural language processing to integrate clinical data for safety evaluations, while CDER’s Information Visualization Platform aids safety reviewers in assessing complex data to identify safety concerns.
Challenges in AI Adoption
AI-driven signal detection holds promise but is hindered by significant challenges: the absence of standardized data sources and formats affects interoperability, the complexity and variability of real-world data may introduce biases, regulatory uncertainty complicates validation and acceptance of AI methods, and concerns about “black box” issues raise questions regarding interpretability and transparency.
Future Directions
To address challenges, healthcare organizations should engage with regulatory bodies for compliance and guidelines, invest in integration strategies with staff training, ensure interpretability through explainable AI techniques, and implement stringent data protection and ethical measures.
CurexBio’s Pharmacovigilance Services
Figure 1 : CurexBio’s Pharmacovigilance Services
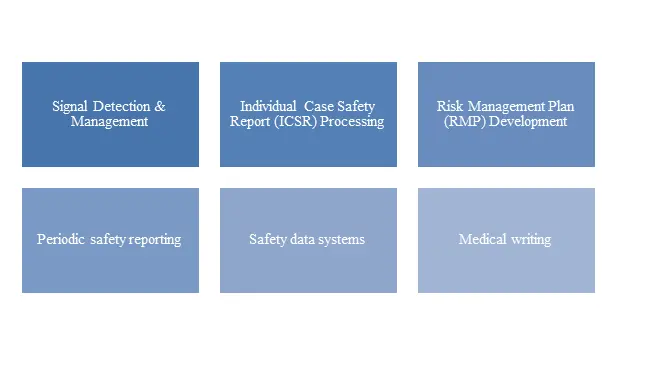
At CurexBio, we offer extensive pharmacovigilance services covering the entire drug safety lifecycle, backed by years of regulatory expertise and operational excellence. To know more about our exclusive pharmacovigilance services, contact us on bd@curexbio.com





