2026 is poised to be a pivotal year in global pharmaceutical regulation, highlighted by significant FDA policy changes and extensive reforms in the EU. These shifts are crucial for pharmaceutical companies, as they can determine the difference between fast-track approvals and extended delays in drug access.
At CurexBio, we assist clients in navigating the evolving regulatory landscape with a focus on pharmacovigilance, clinical operations, and compliance services, operating across India, the USA, and Canada to ensure regulatory excellence.
- FDA
In February 2026, the FDA announced a significant change in its drug approval process, stating that one adequate and well-controlled study, along with confirmatory evidence, will be the standard basis for marketing authorization of new products. FDA commissioner later quoted that “Two trials represent one aspect of clinical credibility, and by 2026, there will be strong alternative methods to ensure product efficacy in improving health and longevity, reducing the need for repetitive manufacturer testing.”
What This Means for Sponsors:
- Approximately 60% of first-of-a-kind drugs have been approved based on single studies in the last five years, indicating that this has become the norm rather than an exception.
- Drugs for common diseases will be significantly impacted, while products for rare diseases and oncology typically already receive approvals based on single trials.
- Confirmatory evidence is essential, prompting sponsors to strategically construct convincing evidence packages that extend beyond just multiple trials.
Table of Contents
ToggleFDA’s “Plausible Mechanism” Framework for Individualized Therapies
On February 23, 2026, the FDA introduced a transformative framework aimed at the development of personalized therapies for ultra-rare diseases, focusing on genome editing and RNA-based therapies like antisense oligonucleotides, with potential applicability to other customized treatments that target the root causes of these diseases.
Key Criteria for the Plausible Mechanism Framework:
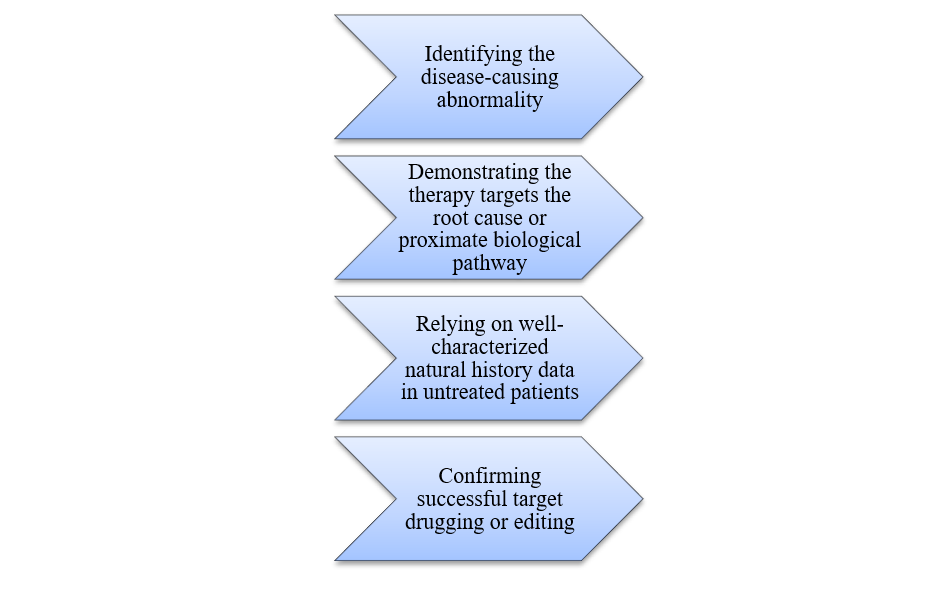
The framework revolutionizes genome editing by allowing a single product application to address multiple mutations in one gene, utilizing master protocols to evaluate product variations in a single trial.
2.EU Pharmaceutical Legislation Overhaul
The EMA Management Board has praised the political agreement on new EU pharmaceutical legislation as a historic milestone, enhancing Europe’s role in pharmaceutical innovation and benefiting patients across the EU.
EMA’s 2026 work programme is guided by a dedicated governance structure involving EMA, the Management Board, the European Commission, and civil society participation throughout key workstreams.
3.ICH E6(R3) Implementation: Global GCP Transformation
Australia’s Therapeutic Goods Administration (TGA) began implementing ICH E6(R3) Good Clinical Practice for clinical trials under the CTN and CTA schemes as of January 13, 2026, with a 12-month transition period allowing compliance with either E6(R2) or E6(R3).
E6(R3) Changes:
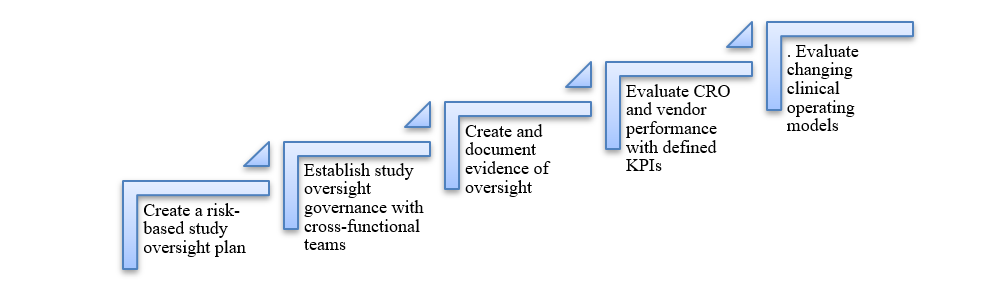
The updated guideline fundamentally redefines clinical trial oversight.

Five Key Steps for Compliance
4.Bayesian Methods Gain Regulatory Recognition
In January 2026, the FDA issued draft guidance regarding the use of Bayesian methodology in clinical trials for drug and biological products, detailing regulatory expectations for Bayesian approaches in trial phases, focusing on adaptive designs and prior information use, particularly when these methods are utilized for primary evidence of safety or effectiveness.
NMPA Implementation: On January 21, 2026, China’s NMPA issued trial implementation guidance on the Guiding Principles for the Application of Bayesian External Information Borrowing Methods in Drug Clinical Trials, promoting the regulated use of existing information in clinical research and advancing Bayesian approaches in drug development.
5.EMA Pediatric Oncology: New Proof-of-Concept Framework
In March 2026, the EMA proposed a concept paper for a future reflection on non-clinical proof-of-concept studies for pediatric oncology treatments, addressing new global requirements, including FDARA amendments that mandate pediatric assessments for oncology products relevant to pediatric cancers.
- Weight-of-Evidence Approach
EMA proposes a weight-of-evidence approach considering various factors.

6.CTD Quality Module Overhaul: ICH M4Q(R2)
On January 20, 2026, the FDA released draft guidance on M4Q(R2) for the Common Technical Document concerning the registration of pharmaceuticals for human use. The update aims to create a globally harmonized framework for organizing and presenting quality data.
7.DARWIN EU Expansion: Real-World Evidence Infrastructure
In 2026, EMA plans to initiate a competitive tender to extend the DARWIN EU project, aimed at enhancing infrastructure for reliable real-world evidence across Europe, from 2027 to 2032.
EMA has initiated over 100 studies since 2022 with 32 data partners covering 188 million patients across 16 European countries. Additionally, it has published its first data strategy for medicines regulation, focusing on governance, quality standards, and stakeholder value delivery.
8.WHO Guidance on Controlled Medicines
The World Health Organization released a guideline on balanced national controlled medicines policies, aimed at ensuring access to controlled drugs for medical purposes while reducing the risks of misuse and diversion.
Key Statistics
Up to 98% of patients in low-income countries lack adequate pain relief, and millions of epilepsy patients remain untreated due to restrictive regulations. The guideline addresses seven domains: policy development, pricing and financing, medicines selection, procurement and supply chain, regulation, prescribing and dispensing, and education.
At CurexBio, we offer comprehensive services to support your regulatory journey, aligning with global regulatory expectations to ensure compliance and competitiveness for your programs.
Our Regulatory-Ready Services
- Pharmacovigilance and safety involves processing Individual Case Safety Reports (ICSRs), submitting regulatory documents such as DSUR, PSUR/PBRER, and PADER/PAER, monitoring literature, evaluating safety data, detecting signals, and providing assistance with EudraVigilance.
- Risk management involves benefit-risk analysis, risk management planning, and proactive safety monitoring to effectively identify and mitigate potential risks.
- Integrated clinical project management involves risk-based monitoring and data management adhering to CDISC standards, along with overseeing sites and vendors.
- Regulatory compliance entails understanding country-specific requirements, maintaining audit-ready reporting, and utilizing an expert safety team.
- Safety Database Management involves configuration and oversight using Oracle Argus or client platforms, ensuring seamless transitions between databases.
At CurexBio, we provide expert support in pharmacovigilance, clinical operations, and regulatory matters to keep your programs proactive rather than reactive. Contact CurexBio today to discuss how our pharmacovigilance, clinical operations, and regulatory services can keep your programs compliant and accelerating through this transformative year.
References:
- FDA Launches Framework for Accelerating Development of Individualized Therapies for Ultra-Rare Diseases.
- WHO publishes full guideline report to help countries ensure safe, equitable access to controlled medicines
- FDA Will Require Only 1 Study to Approve New Drugs, Speeding Up Process
- EMA Management Board: highlights of December 2025 meeting
- Global Regulatory Updates on Clinical Trials (January 2026)
- ICH E6(R3) Unpacked: Diving Deep into the Impacts of the Guideline Changes
- EMA proposes new paper on proof-of-concept studies for pediatric oncology drugs
- M4Q(R2) The Common Technical Document for the Registration of Pharmaceuticals for Human Use: QualityJanuary 2026





