The year 2026 signifies a crucial transition in clinical research, where artificial intelligence has evolved from experimental phases to being integral in operations. It is now a standard practice, with leading organizations embedding AI fluency across all facets of trial execution.
At the SCOPE Summit 2026 in Orlando, attended by over 4,500 clinical research professionals, it was highlighted that AI has transitioned from being experimental to becoming an expectation. AI systems that can think, plan, and act are transforming the design of trials, site selection, patient enrollment, and data management.
Industry leaders anticipate that AI will reduce clinical development timelines by 30%–40% in the next 2–5 years, with the AI in clinical trials market expected to reach $8 billion by 2030, prompting sponsors and CROs to leverage these advancements.
At CurexBio, we leverage innovative technology and scientific expertise across India, USA, and Canada to enhance clinical trial operations, making them faster, smarter, and more reliable.
Table of Contents
ToggleHow AI is impactful in 2026?
- Protocol Design and Feasibility
The challenge: Traditional protocol design is hindered by historical assumptions, resulting in amendments that cause delays in trials and increased costs.
AI Solution: AI-powered platforms allow sponsors to model patient pathways, foresee enrollment issues, and enhance protocol design prior to site activation. Teams are now able to address key questions in advance, including the optimal enrollment plan for completing a study within a designated timeframe, the most cost-effective approach to finalize the study, and strategies to minimize participant burden without compromising scientific rigor.
- Site Identification and Selection
The Challenge: Identifying the right sites globally remains a significant bottleneck in clinical research.
The AI Solution: Semantic knowledge platforms leverage extensive operational data and real-time analytics to achieve exceptional accuracy in matching protocols to sites. The platform will leverage insights from multiple centres to deliver ultra-tailored feasibility assessments
- Patient Recruitment and Retention
The Challenge: Over 50% of clinical trials face delays in enrollment, and maintaining patient retention continues to be a significant challenge.
The AI Solution: AI-powered tools allow for continuous patient matching through adaptive notifications tailored to patient preferences regarding timing and language, as well as conversational electronic Clinical Outcome Assessments (eCOAs) that resemble dynamic interactions rather than static questionnaires. The industry is transitioning from episodic trial management to continuous, adaptive execution, facilitated by AI systems that allow real-time adjustments in recruitment strategies based on changing data.
- Risk-Based Monitoring and Data Integrity
The Challenge: Traditional 100% source data verification is demanding on resources and may not be the most efficient way to utilize monitoring resources.
The AI Solution: AI-powered centralized monitoring facilitates real-time anomaly detection to highlight unusual patterns, predictive risk alerts for early intervention, and automated data cleaning to minimize the need for manual query management. Industry leaders anticipate a future of instantaneous data collection, storage, and cleaning, utilizing AI for mapping source data fields and machine learning to identify patterns beyond human detection.
- Study Planning and Portfolio Optimization
The Challenge: Sponsors must make intricate decisions regarding the allocation of resources across various studies and geographical areas.
The AI Solution: Tools like PSI’s VISIONAL™ integrates historical study data, industry insights, and costing data for real-time modeling of studies and portfolios. It provides full budgets and cash flow projections for various scenarios, facilitating scenario modeling based on specific criteria, understanding of startup timelines and milestone impacts, and optimizing portfolios across therapeutic areas.
- Trial Closeout and Regulatory Submission
The Challenge: Database lock and clinical study report generation are manual and time-consuming processes.
The AI Solution: AI enhances clinical operations by facilitating the generation of clinical study reports, conducting biostatistics and data analysis, preparing SDTM/ADaM datasets, and packaging regulatory submissions. This results in accelerated trial execution and regulatory review, ultimately delivering therapies to patients more swiftly.
The Platformization of Clinical Research
A major trend in 2026 is platformization, which refers to the consolidation of fragmented tools into unified, end-to-end systems, signaling a shift away from piecemeal clinical trial innovation.
- Living Protocols
Traditional protocols are evolving from static PDFs to “living protocols,” which are dynamic, machine-readable documents that allow for continuous simulation, validation, and adaptation. Supported by ICH M11, these protocols facilitate automated data capture, real-time updates, and consistent data reporting across studies. By 2030, the nature of protocols is anticipated to shift from paperwork to software.
- The AI Fluency Divide
A sharp divide is forming between organizations that view AI as an add-on and those that integrate it into all clinical processes. By 2026, AI fluency—assessed through talent, governance, and operational agility—will be crucial for organizational survival.
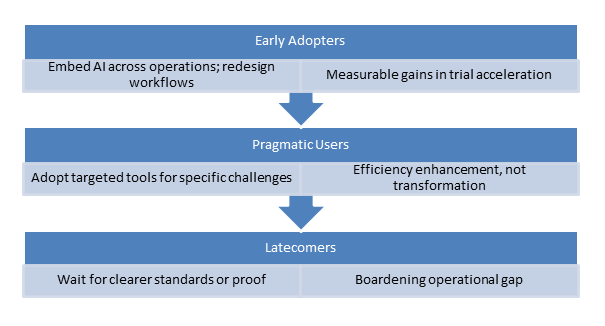
Success hinges on process innovation rather than merely adding technology; organizations need to redesign their workflows instead of simply digitizing current ones.
At CurexBio, we are committed to facilitating transformative clinical trial operations through a blend of technology and a human-centered approach. Our capabilities encompass data management, risk-based monitoring, and comprehensive project management, all rooted in our core values of integrity, innovation, collaboration, and excellence.
Ready to Accelerate Your Clinical Trial with CurexBio? Visit us on bd@curexbio.com





