In clinical research, effective clinical project management is crucial for ensuring trials are completed on time and within budget, distinguishing successful trials from those plagued by delays and cost overruns. Clinical trials involve multiple disciplines and require effective coordination among sponsors, investigative sites, regulatory authorities, data managers, monitors, laboratories, and various stakeholders to avoid derailing the study.
The evolution of clinical project management from a mere support role to a strategic necessity, highlighting its proactive nature. It emphasizes that effective project management involves not just monitoring schedules but also designing success pathways, foreseeing potential challenges, and ensuring all trial components function effectively and meaningfully.
At CurexBio, based in Ahmedabad with operations in India, the USA, and Canada, we excel in project management, driving complex healthcare initiatives efficiently across pharma, biotech, and medical device sectors.
Table of Contents
ToggleWhat Is Clinical Project Management?
Clinical trial project management involves planning, executing, monitoring, and controlling clinical research to safely and effectively introduce new drugs and medical devices. It necessitates collaboration with regulatory bodies, medical professionals, and the clinical trial team while handling various stakeholders, data, timelines, and budgets concurrently.
The discipline of modern clinical project management goes beyond simple task tracking and involves a broader scope of responsibilities and strategies.
Figure 1: CurexBio ensure seamless clinical project management
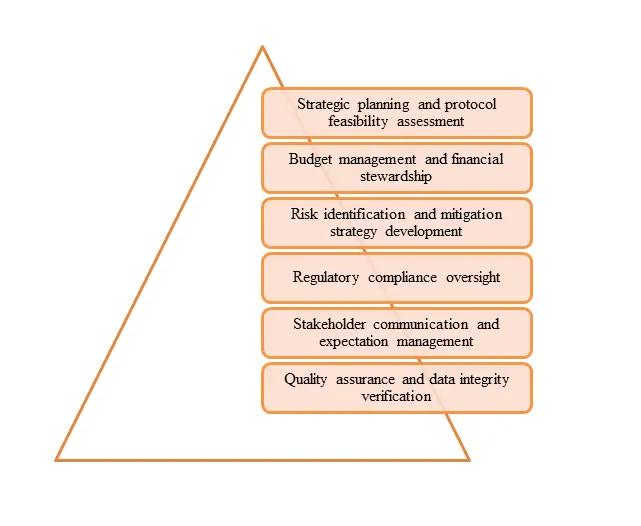
At CurexBio, project managers have extensive therapeutic expertise and experience in all phases of clinical development, from early exploratory studies to large-scale pivotal trials.
Why Clinical Project Management Matters: The Strategic Impact
-
Time bound commitment
The pharmaceutical development process is time-sensitive, with any delay in clinical trials leading to higher costs and postponing patient access to crucial therapies.
- Patient recruitment issues can result in substantial cost overruns due to delays and extra work. A project lead must manage accountability and expectations as study elements change.
- CurexBio approach: Our project managers use advanced tools and methodologies for controlling timelines and budgets, carefully monitoring progress against baselines, identifying variances, and applying corrective actions to maintain schedules and financial parameters, ensuring precision in meeting timelines and milestones.
-
Risk Management and Issue Prevention
- Clinical trials are unpredictable due to factors like staff turnover, enrollment issues, and data discrepancies. The key differentiator between a successful and troubled trial lies in effective proactive risk management.
- CurexBio Approach: Our project managers utilize proactive risk management and mitigation strategies to assess potential threats to timelines, budget, quality, and regulatory compliance. They create contingency plans and implement corrective and preventive actions (CAPAs) to mitigate issues early, thus protecting study integrity and progress. This “risk-proof strategy” allows for the identification and resolution of potential disruptions before they affect study outcomes.
-
Cross-Functional Coordination and Communication
- A clinical trial involves multiple functional groups such as clinical monitoring, data management, biostatistics, regulatory affairs, pharmacovigilance, and medical writing. Centralized coordination is essential to prevent silos, which can lead to misalignment, duplication of efforts, and critical gaps.
- CurexBio Approach :A key aspect of our service is offering a dedicated Project Manager who acts as your single point of contact. This approach enhances communication, maintains consistent messaging, and builds a robust partnership. The Project Manager serves as your advocate within CurexBio, managing all teams to ensure coordination aligns with your strategic goals.
-
Quality and Compliance Assurance
- Regulatory scrutiny of clinical trials has intensified, with health authorities such as the US FDA, EMA, and India’s CDSCO conducting Bioresearch Monitoring inspections to ensure compliance with Good Clinical Practice (GCP) standards. Non-compliance may lead to warning letters, clinical holds, data rejection, and investigator disqualification.
- Our approach: Integrated quality and compliance assurance is a fundamental aspect of our project management framework. Project managers guarantee adherence to the study protocol, Standard Operating Procedures (SOPs), Good Clinical Practice (GCP) guidelines, and relevant regulatory requirements, while collaborating with the Quality Assurance team to ensure audit-readiness and maintain high standards of data integrity and patient safety.
A CurexBio Perspective on the Clinical Project Management Lifecycle
CurexBio supports sponsors through the entire project lifecycle, following the five phases outlined in the Project Management Body of Knowledge (PMBOK): initiation, planning, execution, monitoring and controlling, and closing . Each phase receives dedicated attention and expertise.
- Phase 1: Initiation
- Phase 2: Planning
- The planning phase involves developing detailed roadmaps for scope, schedule, budget, risks, and quality, utilizing tools such as Gantt charts and risk registers to define deliverables and assign responsibilities.
CurexBio Approach: Our project managers engage early in the process to create detailed study plans that encompass timelines, budgets, and resource allocation. They perform feasibility assessments, recognize potential risks, and formulate mitigation strategies to ensure an efficient and successful study startup.
- Phase 3: Execution
- Execution involves implementing the plan through site activation, enrolling patients, collecting data, and coordinating across functions.
CurexBio approach: At CurexBio, project leads manage trials effectively, overseeing training, drug shipment, and patient screening while employing targeted recruitment strategies to tackle challenges such as low enrollment, ensuring timely and scoped execution.
- Phase 4: Monitoring and Controlling
- This phase emphasizes tracking progress, risk management, and quality maintenance during execution, utilizing key performance indicators (KPIs) to assess performance and trigger corrective actions as necessary.
CurexBio approach: CurexBio’s clinical project management integrates centralized and risk-based monitoring to identify issues promptly, uphold data integrity, and guarantee adherence to study protocols. Monitors assess incoming data for errors, inconsistencies, and protocol deviations, producing clear queries and collaborating with site staff for accurate resolution.
- Phase 5: Closing
- The closing phase involves completing all planned project activities, including data lock, final reporting, and archiving documentation, as well as reviewing lessons learned for future improvements.
CurexBio approach: CurexBio assists in closure by handling regulatory filings and documentation, facilitating a smooth transition to post-marketing studies. After trial completion, the project manager finalizes submissions, conducts close-out meetings, and ensures effective knowledge transfer.
Capabilities of CurexBio’s Clinical Project Management
- End-to-End Clinical Program Oversight
CurexBio offers full management of clinical development, encompassing protocol development to post-marketing surveillance, with project managers ensuring alignment with scientific goals, regulatory standards, and patient safety.
- Vendor Coordination and Training
Modern clinical trials are coordinated by project managers at CurexBio, who manage relationships with various external partners, including central laboratories, imaging vendors, and electronic data capture providers, to ensure adherence to specifications and timelines.
- Investigator and Site Team Training
Successful trials rely on well-prepared sites. CurexBio ensures comprehensive training for investigators and site coordinators on protocol requirements, GCP obligations, and data collection procedures.
- Integration with Data Management
Clinical project management at CurexBio closely collaborates with the Data Management team to ensure data quality, cleaning, and reconciliation, thereby supporting timely database lock and regulatory submission.
- Risk-Based Monitoring Oversight
CurexBio utilizes a risk-based approach to enhance efficiency in monitoring while ensuring thorough oversight in Clinical Project Management, Quality & Compliance, and Regulatory Affairs. This strategy prioritizes monitoring on critical risks to data quality and patient safety, thereby optimizing resources without sacrificing oversight.
At CurexBio, we are committed to upholding the trust placed in us for your clinical trial, which embodies years of scientific work and significant financial investment, as well as the hopes of patients awaiting new treatments. Our project managers are skilled and dedicated to ensuring successful outcomes.
Ready to Partner with CurexBio? Contact our Clinical Project Management team to discuss how we can expedite your next clinical trial. CurexBio operates in India, the USA, and Canada, offering a wide range of clinical research services, including protocol development and post-marketing surveillance, to enhance medical research and patient outcomes.





