Table of Contents
ToggleChallenges of Traditional Clinical Data Management in Complex Clinical Studies
For decades, clinical data management (CDM) operated through a predictable process of data entry, batch cleaning, querying, reconciling, and eventual lock after patient visits. However, this linear model faces challenges amid increasing trial complexity, decentralized designs, and demands for real-time regulatory oversight.
A pivotal change is set for 2026, with automation, artificial intelligence, and real-time data integration revolutionizing CDM from a reactive quality check to a proactive function that enhances trial execution. The focus now is on the speed of integrating these technologies into operational practices.
At CurexBio, we are equipped to deliver this future. With operations across nations and other countries, and a team dedicated to innovation and excellence, we provide clinical data management services that leverage cutting-edge automation and AI to deliver cleaner data, faster timelines, and greater regulatory confidence .
The Evolution: From Retrospective to Real-Time
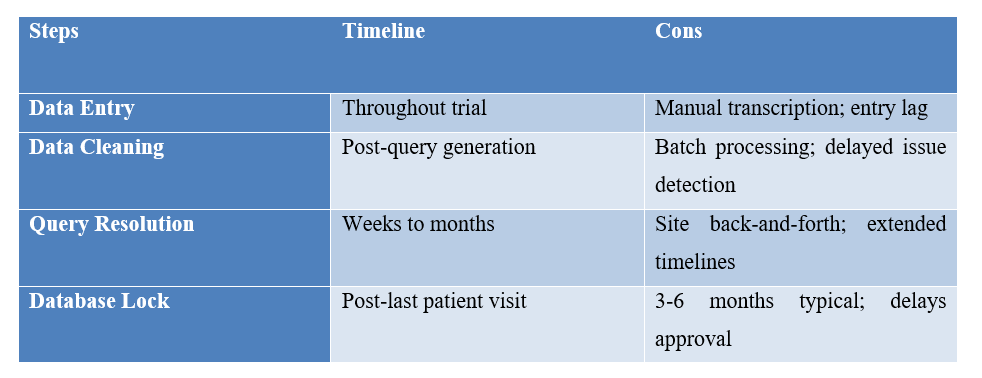
| Steps
|
Timeline | Cons |
| Data Entry | Throughout trial | Manual transcription; entry lag |
| Data Cleaning | Post-query generation | Batch processing; delayed issue detection |
| Query Resolution | Weeks to months | Site back-and-forth; extended timelines |
| Database Lock | Post-last patient visit | 3-6 months typical; delays approval |
This model was effective for smaller, simpler trials at centralized sites, but current global, decentralized, and complex trials require a new approach.
The Emerging Model

Key Trends Shaping Clinical Data Management in 2026
- AI-Powered Data Cleaning and Query Generation
- The Change: Traditional data cleaning requires manual review and query generation, accounting for up to 30% of data management efforts.
- AI in Action:
Figure 1: AI in Action: Machine learning algorithms now automatically
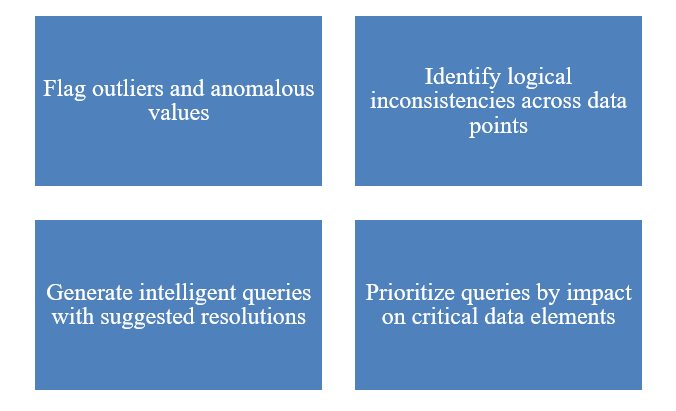
Impact: AI-driven query generation can decrease query volume by 20-40% and enhance query quality, resulting in quicker database locks and less burden on sites for sponsors.
- Risk-Based Data Management
- The Change : ICH E6(R3) promotes risk-based approaches, highlighting that oversight must align with the significance of the data.
Figure 2: How to Implement

- EDC Platform Evolution
- The Change: Electronic Data Capture systems are transforming from basic data entry platforms into sophisticated data hubs.
Figure 3: Modern EDC Capabilities:

-
- Automated edit checks trigger in real-time when data is entered.
- Direct Data Capture (DDC) and eSource
- The Change: DDC eliminates the transcription step found in traditional SDV, which compares CRF entries against source documents.
Figure 4: How it works

EMA and FDA endorse eSource with guidance focusing on data integrity, audit trails, and system validation. CurexBio’s platforms comply with 21 CFR Part 11 and are designed for eSource implementation.
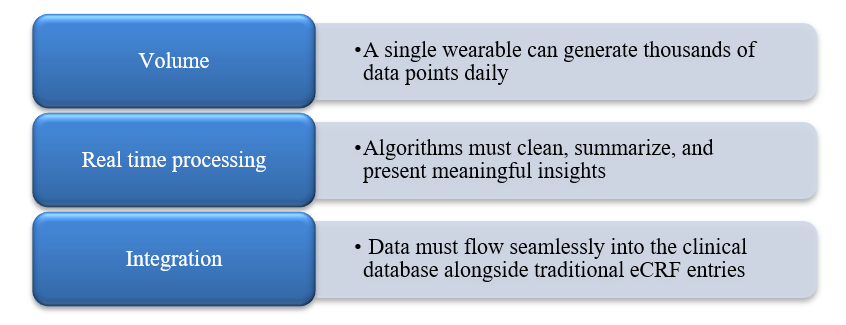
- Wearables and Sensor Integration
- The Change: Continuous physiological data from wearables, smart inhalers, and connected devices is increasingly standard across various therapeutic areas.
Figure 5: Data Management Implications
- Real-World Data Integration
- The Change: Regulators are increasingly accepting real-world data (RWD) for labeling, external controls, and post-marketing commitments.
Figure 6: CDM Considerations

- Generative AI in Data Management
- The Change: Generative AI is progressing from pilot projects to production applications specifically for data management tasks.
Figure 7: Applications:

Industry leaders anticipate that generative AI will cut manual data management efforts by 30-50% in the next 2-3 years.
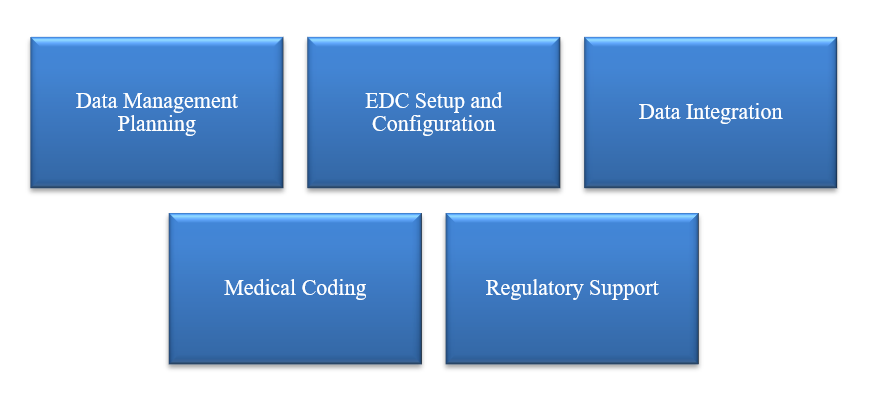
Figure 8: CurexBio supports by Clinical Data Management services