Table of Contents
ToggleChallenges and Innovations in Clinical Trial Patient Recruitment
In clinical research, nearly 80% of clinical trials fail to reach their enrollment targets, with 19% terminated early due to insufficient recruitment. This leads to significant financial losses and delayed access to therapies for patients. However, innovative, evidence-based recruitment strategies, including AI-powered patient matching, decentralized trial designs, and community-focused engagement—are revolutionizing the process. The blog discusses effective patient recruitment strategies and how CurexBio utilizes these methods for timely enrollment and quality data.
Why Recruitment Fails
Understanding the problem is the first step toward solving it.
| Issue | Effect |
| Overly narrow eligibility criteria | High screen‑failure rates; potential participants excluded unnecessarily |
| Burden of frequent site visits | Patients decline to enroll or drop out early |
| Lack of patient awareness | Many eligible individuals never learn about a trial |
| Passive site‑based recruitment | Traditional “wait‑and‑see” approach misses most potential candidates |
| Limited diversity in trial populations | Results may not generalize to real‑world patients |
Site-level pressures in clinical trials are significant, with 35% of sites identifying the complexity of trials as their primary challenge and 30% experiencing staffing shortages.
Proven Strategies That Drive Enrollment
Protocol Design Built for Feasibility
Recruitment success begins before patient contact, with effective sponsors designing protocols that consider real-world patient availability.
- Using real-world data (RWD) is essential for accurately estimating the true eligible patient pool prior to the finalization of inclusion and exclusion criteria.
- Broaden eligibility criteria based on safety, minimizing exclusions related to age, weight, or comorbidities.
- To leverage established FDA precedent protocols while allowing for innovation.
- Conduct evidence-based site feasibility assessments instead of depending on optimistic projections.
A poorly designed protocol leads to slow recruitment, while a protocol based on real-world patient data can significantly speed up enrollment.
Digital Outreach and Social Media Campaigns
Social media has become a central recruitment channel, as evidenced by an analysis of 118 clinical trial recruitment ads from 2025, which indicates that digital outreach is now integrated into active enrollment workflows.
 In a trial for hidradenitis suppurativa, a combination of EHR searches, remote screening, and teledermatology achieved recruitment rates nearly 10 times higher than traditional site-based methods. Digital outreach is effective as it targets potential participants in familiar environments, ensuring that content is clear and compliant with IRB requirements, while being tailored to the language and literacy levels of the target group.
In a trial for hidradenitis suppurativa, a combination of EHR searches, remote screening, and teledermatology achieved recruitment rates nearly 10 times higher than traditional site-based methods. Digital outreach is effective as it targets potential participants in familiar environments, ensuring that content is clear and compliant with IRB requirements, while being tailored to the language and literacy levels of the target group.
AI‑Powered Patient Identification and Matching
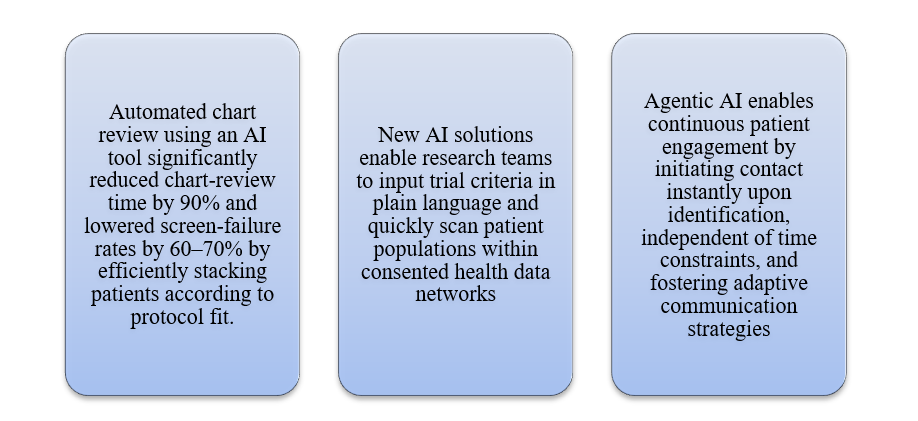
Artificial intelligence is revolutionizing recruitment by shifting it from a reactive to a proactive and predictive process. Modern AI systems can process extensive unstructured clinical data to identify potential eligibility patterns that might be overlooked.
Clinical trials often fail not due to a lack of participant willingness, but because the majority of potential candidates are not identified. AI has the potential to resolve this issue.
Patient‑Centricity and Reduced Burden

Modern recruitment emphasizes treating patients as volunteers rather than subjects; strategies that alleviate logistical and psychological burdens are more effective.
Research by the PACT Consortium indicates that decentralized clinical trial (DCT) components lead to faster enrollment timelines and reduced screen-failure rates. Additionally, DCTs enhance participant diversity, with Asian enrollment increasing from 14.2% to 20.9% and American Indian or Alaska Native participation nearly quadrupling from 0.5% to 1.9%.
Diversity and Community Engagement

The FDA’s December 2025 guidance advises early engagement with patient advocacy groups, utilizing real-world data, and choosing research sites in diverse geographic areas.
Diversity is both an ethical and scientific necessity, as treatments effective only in limited populations can exclude many patients.
Site Empowerment and Responsive PI Selection
Sites play a crucial role in recruitment, and effective sponsors provide necessary support to them:
Ongoing communication ensures site readiness and addresses barriers in real time. Responsive principal investigator selection focuses on PIs who are enthusiastic about the trial and effectively communicate this to patients. Recruitment is phased to scale site activation based on actual performance instead of initial projections, coupled with a reduction of administrative burden to allow site staff to concentrate on patient engagement.
A single motivated research coordinator, equipped with appropriate tools, can achieve better results than a large academic institution with numerous staff members.
Measuring What Matters

Effective recruitment involves ongoing monitoring and adjustment to optimize hiring processes.
Effective patient recruitment is a comprehensive system that begins with protocol design focused on real-world feasibility and incorporates digital outreach, AI-powered identification, and patient-centric logistics, while emphasizing diversity through community engagement and providing responsive management support to sites.
Sponsors and CROs that implement evidence-backed strategies consistently experience positive outcomes.

At CurexBio, recruitment is integrated into trial operations from the outset, employing a patient-centric and data-driven strategy.
Our Capabilities:
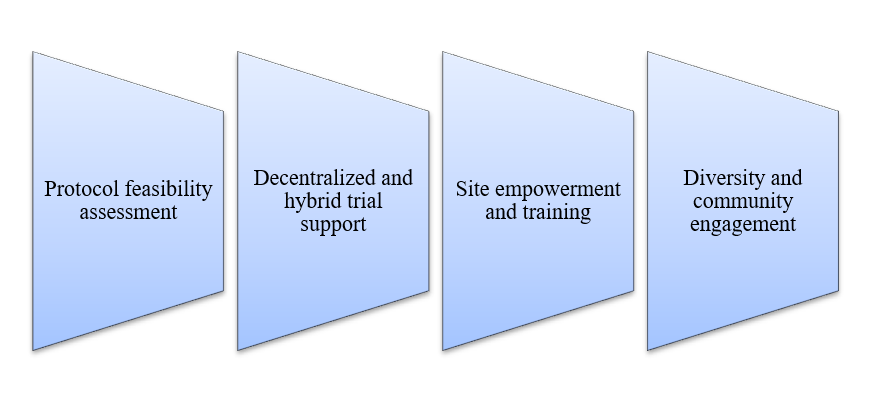 With operations nationally and globally, CurexBio emphasizes quality over quantity, undertaking fewer projects to ensure exceptional results for clients. Our integrated teams support patient recruitment from Phase I through post-marketing surveillance, focusing on driving scientific progress. To enhance your patient recruitment strategy, contact CurexBio for evidence-backed methods to accelerate your clinical trial.
With operations nationally and globally, CurexBio emphasizes quality over quantity, undertaking fewer projects to ensure exceptional results for clients. Our integrated teams support patient recruitment from Phase I through post-marketing surveillance, focusing on driving scientific progress. To enhance your patient recruitment strategy, contact CurexBio for evidence-backed methods to accelerate your clinical trial.
References:
- Trial of SiTes to IncreAse diversity in clinical TriaLs (TOTAL): A study protocol paper
- New FDA Guidance: Further Enhancing Clinical Trial Participation https://www.emvenio.com/news-and-resources/new-fda-guidance-clinical-trial-participation/
- Scaling Recruitment Across Therapeutic Areas and Regions https://www.appliedclinicaltrialsonline.com/view/scaling-recruitment-across-therapeutic-areas-regions
- Clinical trial patient recruitement on social media: Trends, Strategies, and Real Compaign Data https://trialx.com/clinical-trial-patient-recruitment-on-social-media/
- Go Digital: Smart Strategies for Clinical Trial Patient Recruitment https://lifebit.ai/blog/digital-patient-recruitment-complete-guide/
- What Really Drives Recruitment, Enrollment, And Retention — According To A PI
- https://www.lindushealth.com/blog/what-really-drives-recruitment-enrollment-and-retention-according-to-a-pi?f32c9f01_page=5
- 2025 Highlights: Impact in Action-Advancing Patient Recruitment Management & Remote Data Collection with AI https://trialx.com/2025-highlights-impact-in-action-advancing-patient-recruitment-management-remote-data-collection-with-ai/





