The pharmaceutical industry is facing significant challenges with rising drug development costs exceeding $2.6 billion per therapy and prolonged clinical trial timelines. With internal R&D teams becoming leaner and regulatory complexities increasing, there is a pressing need for quicker breakthroughs in treatments. It is evident that, as of 2026, no single pharma company can operate independently; strategic outsourcing is essential for gaining a competitive edge, rather than merely serving as a cost-reduction strategy.
Table of Contents
ToggleThe Shifting Landscape: Why In-House Everything No Longer Works
Large pharmaceutical companies historically relied on a vertical integration model, encompassing discovery, development, manufacturing, and commercialization within a single entity; this approach, however, is now proving unsustainable.
- Rising complexity: Precision medicines, gene therapies, and rare disease trials necessitate specialized expertise that is often lacking in internal teams, as each modality requires unique operational playbooks, regulatory strategies, and site relationships.
- Globalization : Running trials in over 40 countries necessitates understanding local ethics boards, patient recruitment practices, and regulatory nuances, which can take years and substantial financial investment to develop internally.
- Talent Shortage: Clinical research professionals, including CRAs and biostatisticians, are in high demand, with even large pharmaceutical companies facing challenges in filling critical roles across their study portfolios.
- Technology acceleration: AI-driven patient identification, decentralized trial platforms, and real-world data integration necessitate ongoing investment in tools that are often not prioritized for development or maintenance by pharmaceutical organizations.
Outsourcing clinical development to specialized partners enables pharmaceutical companies to concentrate on breakthrough science, while experts handle operational execution.
5 Reasons Outsourcing is the Future of Clinical Development
Speed to Market
In the pharmaceutical industry, timely drug launches are crucial, as a month-long delay can lead to over $100 million in lost revenue. Outsourced Contract Research Organizations (CROs) expedite this process by providing established infrastructure, site networks, and efficient procedures, allowing for quicker startup timelines, faster site enrollment, and effective patient recruitment tailored to specific indications.
Access to Specialized Expertise
No internal team can be knowledgeable in all areas; therefore, outsourcing to a CRO with expertise in specific fields, such as cell therapy trials or decentralized trials involving wearables, allows pharma companies to leverage specialized knowledge without the burden of permanent headcount.
Risk Mitigation and Regulatory Navigation
Regulatory expectations are continuously evolving, particularly with the FDA experiencing capacity constraints in 2025–2026 and global harmonization being inconsistent. CROs employ regulatory affairs teams to monitor these changes, adeptly managing submission structures, advisory committee preparations, and addressing new requirements such as the EMA-FDA AI guiding principles. Outsourcing provides access to valuable institutional knowledge.
Flexible and Scalable Capacity
Pharma pipelines are characterized by their unpredictability, where a drug may progress quickly through phases or encounter safety issues that require changes. Outsourcing to Contract Research Organizations (CROs) provides flexibility for sponsors to adjust resource needs without staff changes, whether in increasing monitors temporarily or pausing studies while maintaining site relationships.
Cost Efficiency Without Compromising Quality
Outsourcing in pharmaceuticals enhances capital allocation and quality of data. By utilizing CROs, companies avoid the high costs of global trial infrastructure and benefit from economies of scale, leading to better data quality at reduced per-patient costs.
Outsourcing in the CRO industry has evolved from a “commodity” model focused on low bids to strategic partnerships. In these partnerships, CROs act as extensions of sponsors’ teams, sharing risk and aligning on long-term goals. They co-invest in innovation and contribute ideas that enhance timelines and patient outcomes, ultimately aiding in the design of more effective study protocols.
Why CurexBio is the Right Outsourcing Partner for the Future
CurexBio is a clinical development partner designed for the needs of 2026 and beyond, distinguishing itself from other CROs.
CurexBio specializes in complex therapeutic areas such as oncology, rare diseases, neurology, and every other therapeutic areas leveraging AI for patient recruitment and data monitoring while ensuring a human-centric approach through Clinical Research Associates (CRAs) and project managers. We operates globally excelling in local regulatory and site management to enhance startup speed and patient retention.
We offers flexible engagement models, from full-service development to functional support, tailored to client needs. CurexBio’s unified clinical platform provides 24/7 access to real-time metrics, fostering transparency and actionable insights. We emphasize partnership over a vendor mentality by assigning dedicated strategic advisors who collaborate closely with clients to ensure shared success.
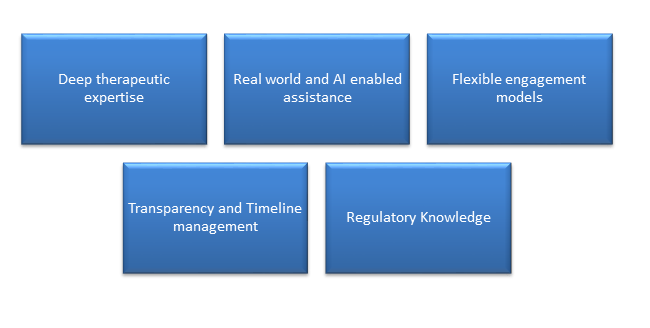
CurexBio is ready to be that partner. Expertise and technology tailored to support both small biotech firms launching pivotal trials and global pharma companies optimizing development portfolios.
Ready to see how CurexBio can accelerate your clinical development program while reducing risk and cost? Contact us today for a consultation. Together, we’ll design a partnership that delivers.





