For decades, India has been recognized as the “pharmacy of the world,” supplying over 20% of global generic medicines and significantly impacting markets like the U.S. and antiretrovirals. However, by 2026, India aims to evolve beyond being a supplier of off-patent medicines. A combination of government policies, regulatory reforms, and private investments is transforming the country into a center for drug innovation, with developed clinical development services facilitating the transition from scientific discovery to approved therapies.
This blog discusses the transformative impact of India’s clinical development services on drug innovation, the underlying factors propelling this shift, and its implications for global pharmaceutical research and development (R&D).
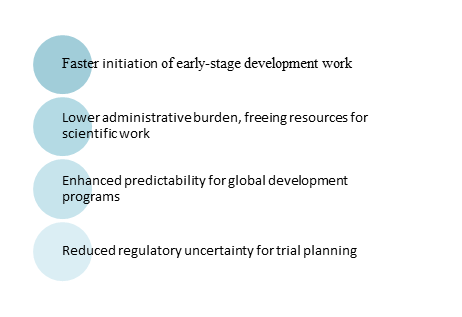
The key catalyst for India’s clinical research transformation is regulatory reform, exemplified by the January 28, 2026 amendments to the New Drugs and Clinical Trials (NDCT) Rules, 2019, which significantly expedite clinical development processes.
Table of Contents
ToggleKey Reforms at a Glance
| Reform | Previous Rule | New Rule | Impact |
| Test licence for non-commercial drug manufacture | Prior approval required | Prior intimation only | 90+ days saved |
| Processing timeline for test licence applications | 90 working days | 45 working days | 50% reduction |
| Low-risk BA/BE studies | Prior permission required | Online intimation | Immediate commencement |
The government’s analysis indicates that proposed reforms will reduce drug development timelines by at least 90 days, significantly benefiting approximately 30,000 to 35,000 test licence applications and 4,000 to 4,500 BA/BE study applications processed annually by the Central Drugs Standard Control Organisation (CDSCO).
What the Reforms Mean for Innovation
The transition from “prior approval” to “prior intimation” indicates a significant shift in regulatory philosophy, showcasing the government’s commitment to trust-based regulatory reforms, specifically reflecting the Jan Vishwas Siddhant (principle of trust). This change is particularly relevant for pharmaceutical innovators.
Reforms in India’s regulatory environment keep safety safeguards for high-risk categories like cytotoxic drugs, narcotics, and psychotropic substances, which still require prior approval. However, for most development work, the regulatory framework has become one of the most enabling globally.
The Biopharma SHAKTI Programme
Regulatory reform is insufficient for creating an innovation ecosystem, which also necessitates ongoing investment, infrastructure, and talent. The Biopharma SHAKTI (Strategy for Healthcare Advancement through Knowledge, Technology & Innovation) programme, introduced in the Union Budget 2026 with a budget of ₹10,000 crore over five years, aims to provide these essential components.
Pillars of the SHAKTI Programme
- 1,000+ Accredited Clinical Trial Sites
The government plans to establish over 1,000 accredited clinical trial sites in India to enhance clinical research quality, speed up drug development, improve patient access to innovative therapies, and create opportunities for researchers and medical professionals. This initiative significantly scales India’s clinical research capacity, making it a key location for global trials.
- Strengthened Regulatory Capacity
The CDSCO will be strengthened by creating a dedicated scientific review cadre and specialists to meet global standards and approval timeframes, addressing global sponsors’ concerns about regulatory predictability and scientific expertise.
- Expanded Pharmaceutical Education
Three new NIPERs will be established, and seven existing institutes will be upgraded, ensuring India’s clinical development services have skilled professionals for complex, innovative trials.
What This Means for Innovation
The integration of government-led site expansion and private-sector training initiatives is fostering a positive feedback loop. This cycle enhances the number of trained investigators and clinical sites, attracting more global trials, which further develops expertise and infrastructure. This infrastructure plays a vital role in driving innovation by offering the clinical validation capacity essential for biopharmaceutical companies to progress new therapies.
The Evolution of Indian CROs: From Generics to Innovation
Indian Contract Research Organizations (CROs) in India have significantly transformed since the early 1990s. Initially concentrating on bioequivalence studies for generics and clinical trials, they have expanded their services to include preclinical offerings and now provide comprehensive, end-to-end solutions throughout the drug discovery, development, and manufacturing continuum.
Key Capabilities of Modern Indian CROs
| Service Area
|
Evolved Capability |
| Discovery | Target identification; lead optimization; proprietary chemistry platforms |
| Preclinical | GLP toxicology; pharmacokinetics; safety pharmacology |
| Clinical | Phase I-IV trials across multiple therapeutic areas; global multi-center management |
| Manufacturing | CMC development; scale-up; commercial manufacturing |
| Biologics | Analytical characterization; cell line development; clinical bioanalysis of large molecules |
Strategies for Sustaining Innovation
Industry leaders have outlined strategies to help Indian Contract Research Organizations (CROs) maintain their innovation momentum.

The Talent Imperative: Building India’s R&D Workforce
India’s clinical development services sector needs a skilled workforce proficient in therapeutic modalities, regulatory science, and digital technologies for innovation to occur.
The NIPER Expansion
The establishment of three new NIPERs and the upgradation of seven existing ones signifies a strong commitment to pharmaceutical education and research, fostering advanced scientific education and skills development, which is crucial for innovation-led growth, as highlighted by Satish Reddy.
Manthan 2026: A Platform for Talent Development
The Manthan 2026symposium focused on enhancing India’s life sciences R&D ecosystem through strong collaboration, patient-centric research models, regulatory alignment, and skill development. Discussions emphasized the need for talent development to match infrastructure investments for achieving innovation goals.
The Global Context: Why India Now?
India’s clinical development services are rising at a moment of significant global opportunity.
-
Supply Chain Diversification
Global pharmaceutical companies are pursuing a “China+1” strategy to diversify their R&D and manufacturing operations, leading to a significant demand for alternative locations. India is well-positioned to meet this demand due to its scale, scientific capability, and cost competitiveness.
-
Cost-Effective Innovation
India provides high-quality clinical development services at costs 30-50% lower than Western markets, ensuring scientific rigor, which is particularly beneficial for cash-strapped biotech startups and cost-sensitive pharmaceutical companies.
-
Access to Diverse Patient Populations
India’s genetic diversity, extensive treatment-naïve populations, and significant burden of diseases such as diabetes, cardiovascular disease, and cancer position it as an optimal site for trials needing quick enrollment and varied patient groups.
-
Aligned with Global Standards
India is now recognized as a legitimate partner in global drug development due to enhanced regulatory capacity, GCP-aligned investigator training, and CDSCO approvals acknowledged by global health authorities.
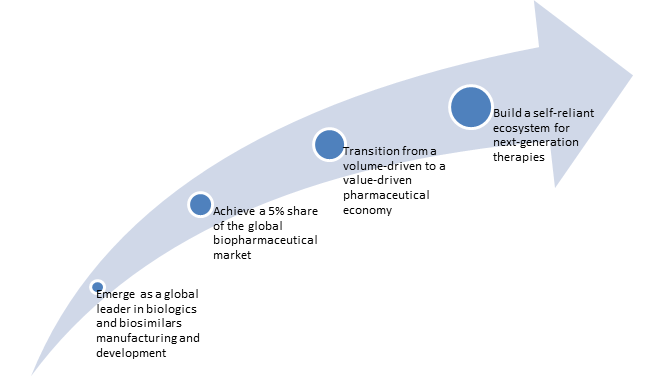
Future Outlook: From Volume to Value Leadership
The transformation of India’s clinical development services aims to fundamentally change the country’s contribution to global health, beyond just increasing its share of the global CRO market.
- Near-Term Priorities (2026-2028)

- Long-Term Vision (2030-2047)
As articulated by the government’s “Viksit Bharat 2047” vision, India aims to:
For global pharmaceutical companies, partnering with CurexBio offers scale, scientific depth, cost efficiency, and a commitment to innovation, enabling faster access to breakthrough therapies for Indian patients and facilitating India’s shift from generics to innovation leadership. CurexBio provides expertise in clinical development, regulatory affairs, and GxP compliance, assisting sponsors through India’s evolving landscape from IND to market approval.





