In clinical research, data serves as the essential foundation for regulatory decisions, therapy approvals, and patient outcomes. Each data point reflects a patient’s experience, safety signals, or efficacy measures, and the integrity of this data is crucial for ensuring that effective therapies are available to those in need.
Clinical trial monitoring is essential for ensuring that trials adhere to protocols, SOPs, GCP, and regulatory requirements. It safeguards the integrity of the data collected, protecting patients and maintaining the potential for regulatory approval. Without thorough monitoring, trials may yield unreliable outcomes.
At CurexBio, our clinical monitoring practice emphasizes quality and compliance, operating in India, USA, and Canada. We provide comprehensive monitoring services to ensure that trial data meets regulatory standards. This blog discusses the importance of clinical trial monitoring for maintaining data integrity and compliance, highlighting how CurexBio’s approach addresses these needs effectively.
Table of Contents
ToggleThe Regulatory Agencies associated Expectations for Data Integrity
Regulatory agencies globally are increasingly focusing on data integrity in clinical trials, with the FDA, EMA, and ICH updating their guidelines to address the evolving landscape of digital, decentralized, and technology-enabled research.
Key Regulatory Developments
| Agency
|
Guidance | Key Requirements |
| FDA | 2024 Guidance on Electronic Systems | Document system requirements; map clinical data flow from capture to archival; adopt risk-based validation |
| EMA | 2023 Guideline on Computerized Systems | Structured system inventories; ALCOA++ compliance; audit trail reviews; direct regulatory access to records |
| ICH | E6(R3) Draft | Data governance frameworks; fitness-for-purpose assessments; clear definition of responsibilities across parties |
These regulations emphasize the need for sponsors to ensure transparency and control throughout the entire lifecycle of clinical data, with monitoring serving as the key method for achieving this.
The ALCOA+ Framework

Data integrity is based on the ALCOA+ principles, serving as a benchmark for regulatory inspectors.
Clinical monitoring verifies each of these principles in practice, ensuring that data entering the trial database meets regulatory expectations .
How Monitoring Ensures Data Integrity
Data integrity is systematically achieved through oversight, as demonstrated by CurexBio’s comprehensive monitoring approach that addresses it across multiple dimensions.
- Source Data Verification (SDV)
The cornerstone of data integrity is Source Data Verification (SDV), which involves comparing data entered into the Case Report Form (CRF) or Electronic Data Capture (EDC) system against original source documents. Monitoring focuses on ensuring that informed consent forms, eligibility criteria, adverse events, concomitant medications, and visit dates all match the supporting documentation. SDV is crucial for identifying transcription errors, such as misplaced decimal points or transposed values, before the data becomes part of the locked database.
- Audit Trail Review
Modern clinical trials utilize electronic systems that necessitate audit trails—secure records documenting all data-related events. Key components of audit trails include timestamps, user identification, change descriptions, original and new values, and modification reasons. Regulatory requirements mandate audit trail reviews, with monitors being vital in this process. Audit trails serve not only as regulatory compliance measures but also as tools for investigating arising issues.
- Investigational Product Accountability
Data integrity in handling investigational products involves monitoring proper storage conditions, accurate dispensing and return records, maintaining chain of custody documentation, and reconciling used and unused products. Discrepancies in product accountability may suggest broader data integrity concerns and potentially lead to regulatory scrutiny.
- Protocol Adherence Monitoring
Accurate data is rendered useless if derived from patients not meeting eligibility criteria or if procedures diverge from the protocol. Monitors evaluate compliance with inclusion/exclusion criteria, adherence to scheduled assessments, proper execution of study procedures, and documentation of protocol deviations.
- Proactive Query Resolution
Monitors detect real-time discrepancies, generate queries, and collaborate with site staff for accurate resolution, reducing post-database lock data cleaning and ensuring a dataset ready for analysis.
The Link Between Monitoring and Regulatory Compliance
Regulatory compliance is evidenced through documentation showing proper trial conduct, with monitoring being a key contributor to this evidence.
- Inspection Readiness
Regulatory inspections can happen anytime, whether during trials, post-database lock, or long after completion. CurexBio maintains inspection readiness through a robust monitoring approach.
Monitors ensure Investigator Site Files (ISF) contain present, current, and properly organized regulatory documents.
- Adverse events and protocol deviations are to be documented and reported within specified timelines.
- Site personnel training is verified and documented.
- Monitoring visit reports provide thorough documentation of all monitoring activities, establishing a comprehensive audit trail for oversight.
Common Inspection Findings Related to Monitoring

Regulatory authorities frequently cite monitoring deficiencies in warning letters and Form 483 observations:
Each of these findings can be prevented through rigorous monitoring.
- Regulatory Endorsement
Regulatory guidance from FDA, EMA, and ICH endorses risk-based approaches, with the ICH E6(R2) guideline highlighting that centralized monitoring provides significant advantages over traditional methods across a wider array of trials.
CurexBio’s Clinical Monitoring Services
CurexBio offers comprehensive clinical monitoring services for all phases of clinical development with operations in India, the USA, and Canada, supported by a team of experienced Clinical Research Associates (CRAs) to ensure effective trial oversight.
Figure 1: CurexBio’s Clinical Monitoring Services
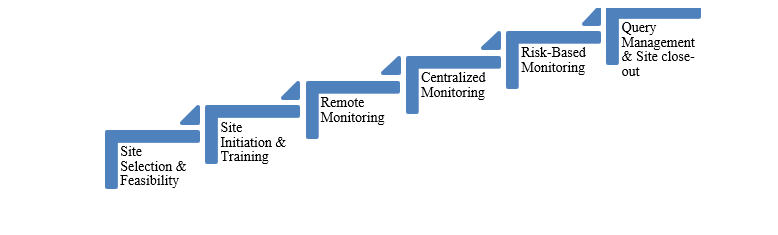
At CurexBio, our monitoring expertise ensures vigilant oversight of your data, patients, and regulatory success from site selection to close-out. CurexBio offers comprehensive clinical monitoring services for all phases of clinical development with operations in India, the USA, and Canada, supported by a team of experienced Clinical Research Associates (CRAs) to ensure effective trial oversight. Contact CurexBio’s Clinical Monitoring Team to enhance your trial oversight, ensuring the integrity of clinical data and successful regulatory submissions.





